Chimie 1
Section outline
-
Teacher: BOUKHATEM Horiya
Department: Physics
Teaching unit: Fundamental Unit
Subject: Chemistry 1
Coefficient: 03
Credits: 06
Evaluation method: Continuous Assessment: 33%, Exam: 67%
-
This “Chemistry 1” course is aimed to L1 Material Sciences (SM) students. It is made up of five chapters.
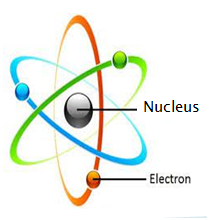
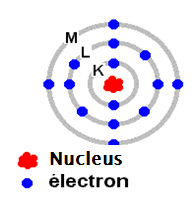
The first chapter is devoted to the study of the structure of the atom (highlighting the proton, neutron and electron), the symbolic representation of the nucleus of an atom and the bonding energy.
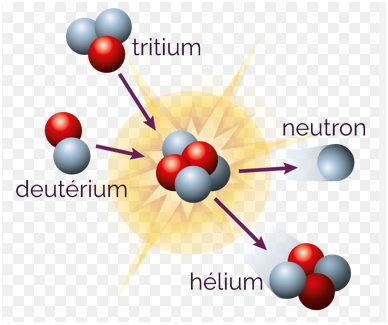
The second chapter covers the study of radioactivity (natural and artificial), the different types of radioactivity (fission, fusion, transmutation, α, β and γ) and the kinetic and energetic study of nuclear disintegrations.
In the third chapter, we will see the photoelectric effect, the hydrogen emission spectrum, the Bohr model, Heisenberg uncertainty principle, Schrödinger equation, quantum numbers as well as the rules for filling atomic orbitals.
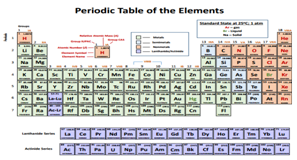
The fourth chapter represents the different construction rules and the periodic classification of chemical elements (periods, groups, families, blocks, physical and chemical properties of the elements).
The last chapter describes the different types of chemical bonds, the Lewis model and dipole moment, the energy diagrams of homonuclear and heteronuclear diatomic molecules, the types of hybridization and the Gillespie's rule.
-
To advance in this course, prior mathematical knowledge of integrals and logarithm and exponential functions is necessary.
-
Program
Chapter 1 : Main components of matter
Chapter 2 : Radioactivity – Nuclear reactions
Chapter 3 : Electronic structure of the atom
Chapter 4 : Periodic classification of elements
Chapter 5 : Chemical bonds
-
[1] R. Ouahès et B. Dévallez, « Chimie générale », Editions OPU - Alger, 04-1993.
[2] F. Rouquérol, G. Chambaud, R. Lissillour, « Exercices résolus de chimie physique », 3e édition, © Dunod, Paris, 2008.
[3] N. Benosmane, « Cours Structure de la Matière (Chimie 1) », Université M’Hamed Bougara (UMBB), 2016/2017.
[4] N. Bendaoud, « Structure de la matière Chimie 1- Cours & Exercices », Université Mohammed Boudiaf d’Oran, 2015/2016.
[5] N. Brinis – Chilla, « Polycopié de Cours-Structure de la matière », Université A. Mira –Bejaia, 2017/2018.